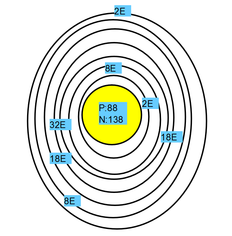
This is a bohr model of of a Radium atom
Facts about the element Radium
General Properties: Radium's symbol is Ra and it's atomic number is 88, it's average atomic mass is 226.0254. It is the heaviest alkali earth metal (group 2) and is in period 7. Radium has 7 energy shells, it's electron configuration is 2, 8, 18, 32, 18, 8, 2. It also has 2 valence electrons. Radium Chloride, Radium Bromide, Radium Hydroxide and Radium nitrate are the compounds associated with Radium.
Physical Properties: Radium is a white silvery solid metal in it's pure form. The density of Radium is 5.5 g/cm3 (cubed). It's melting point is at 700 degrees Celsius, 973 degrees in Kelvin and 1,292 degrees Fahrenheit. Radium's boiling point is 1737 degrees Celsius, 2,010 degrees in Kelvin and 3,158.6 degrees Fahrenheit. The heat of vaporization for Radium is exactly 37 Kj/mol.
Chemical Properties: The element Radium is radioactive and has a half life of 1,601 which then becomes radon gas. Radium is almost a pure white solid alkali earth metal but oxidizes to the exposure of air and becomes a black color. When exposed to air around it, it reacts violently with the air forming radium nitride. All of Radium's isotopes are highly radioactive and there are 25 different known isotopes, 4 of which are found in nature.
Cost: Radium dose not come in large quantities in mines, there for it is not really abundant. Per gram, Radium is about 100$ in it's pure state.
Glossary: Kj/mol (or Kilo-joule per mole) is the SI derived unit for energy per amount of material.
Physical Properties: Radium is a white silvery solid metal in it's pure form. The density of Radium is 5.5 g/cm3 (cubed). It's melting point is at 700 degrees Celsius, 973 degrees in Kelvin and 1,292 degrees Fahrenheit. Radium's boiling point is 1737 degrees Celsius, 2,010 degrees in Kelvin and 3,158.6 degrees Fahrenheit. The heat of vaporization for Radium is exactly 37 Kj/mol.
Chemical Properties: The element Radium is radioactive and has a half life of 1,601 which then becomes radon gas. Radium is almost a pure white solid alkali earth metal but oxidizes to the exposure of air and becomes a black color. When exposed to air around it, it reacts violently with the air forming radium nitride. All of Radium's isotopes are highly radioactive and there are 25 different known isotopes, 4 of which are found in nature.
Cost: Radium dose not come in large quantities in mines, there for it is not really abundant. Per gram, Radium is about 100$ in it's pure state.
Glossary: Kj/mol (or Kilo-joule per mole) is the SI derived unit for energy per amount of material.
